

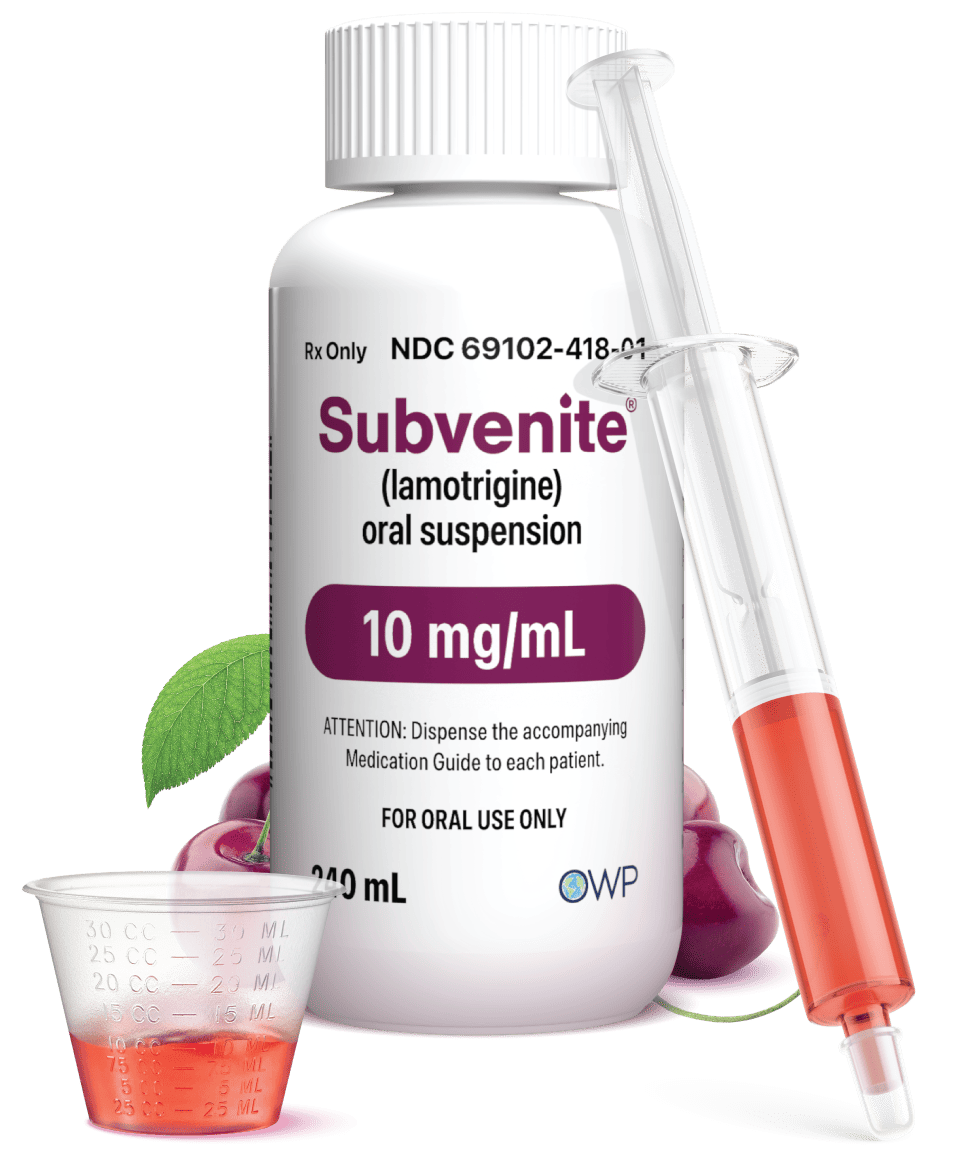
NOW AVAILABLE
The ONLY FDA-Approved Ready-to-Use* lamotrigine oral suspension
*Shake well before use
- Individualized dosing during titration of lamotrigine
- Flexibility as patients progress in their treatment path
- No compounding or crushing needed
- Available at most retail pharmacy within 24 hours
Talk with your prescribing healthcare provider to see if SUBVENITE® (lamotrigine) Oral Suspension is right for you.
For What Conditions is SUBVENITE Oral Suspension Indicated?
SUBVENITE is indicated for:
Epilepsy—adjunctive therapy in patients aged 2 years and older:
- partial-onset seizures.
- primary generalized tonic-clonic seizures.
- generalized seizures of Lennox-Gastaut syndrome.
Epilepsy—monotherapy in patients aged 16 years and older: Conversion to monotherapy in patients with partial-onset seizures who are receiving treatment with carbamazepine, phenytoin, phenobarbital, primidone, or valproate as the single antiepileptic drug.
Bipolar disorder: Maintenance treatment of bipolar I disorder to delay the time to occurrence of mood episodes in patients treated for acute mood episodes with standard therapy.

For What Conditions is SUBVENITE Oral Suspension Indicated?
SUBVENITE is indicated for:
Epilepsy—adjunctive therapy in patients aged 2 years and older:
- partial-onset seizures.
- primary generalized tonic-clonic seizures.
- generalized seizures of Lennox-Gastaut syndrome.
Epilepsy—monotherapy in patients aged 16 years and older: Conversion to monotherapy in patients with partial-onset seizures who are receiving treatment with carbamazepine, phenytoin, phenobarbital, primidone, or valproate as the single antiepileptic drug.
Bipolar disorder: Maintenance treatment of bipolar I disorder to delay the time to occurrence of mood episodes in patients treated for acute mood episodes with standard therapy.
Start Saving on SUBVENITE® (lamotrigine) Oral Suspension
Eligible patients* present this Savings Card (either printed OR on a mobile device) to pay as little as $0.
*Program Restrictions: Not eligible if prescriptions are paid by any state or federally funded programs, including, but not limited to Medicare, Medicaid, VA, DOD, Tricare, or Medigap. Void where prohibited by law. Not valid for an OWP prescription reimbursed in full by any third-party payer. May not be combined with any other coupon, discount, savings card, or other offer. May not be accepted at all pharmacies. Limited to one prescription per person for any 35-day period. No substitutions permitted.

SUBVENITE® (lamotrigine) Oral Suspension is widely available at most retail pharmacies within 24 hours
For general questions or inquiries please contact us at: info@owppharma.com. For SUBVENITE (lamotrigine) Oral Suspension medical questions please contact us at: medinfo.OWP@apcerls.com or call 1-800-273-6729 and follow the prompts for medical information requests. For SUBVENITE (lamotrigine) Oral Suspension safety-related concerns please contact us at: safety.OWP@apcerls.com or call 1-800-273-6729 and follow the prompts for medical adverse events reporting.
SUBOS1013V2 2/26

